VASCADE MVP®
Early Ambulation. Simple. Proven.
VASCADE MVP® venous vascular closure system provides rapid haemostasis for electrophysiology (EP) procedures, enabling patients to get up earlier. VASCADE MVP system is easy to use and has demonstrated 0% major complicationsa in 1,223 patients in 5 EP clinical trials.1-5 In the AMBULATE pivotal trial1, VASCADE MVP system has been proven to reduce time to ambulation by 64%, improve patient satisfaction by 63% and reduce opioid use post procedure by 58%. The VASCADE MVP system is indicated for use in multi-stick and multi-limb catheter-based procedures.6
aMajor venous access site closure-related complications through the follow-up period.
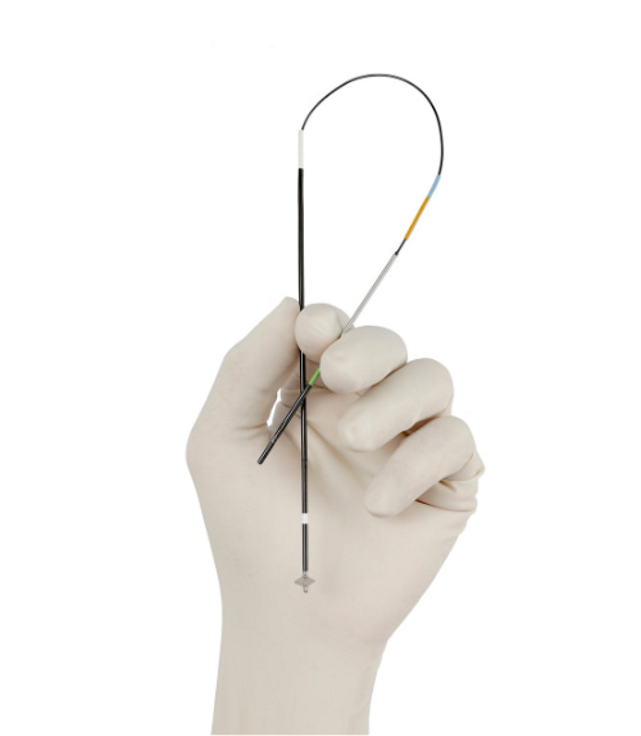
VASCADE MVP® System
Make a selection to learn more
VASCADE MVP® System
3.9 hour reduction in median time to ambulation
Ordering Information
|
AMBULATE Trial
|
The AMBULATE clinical study has shown that the use of VASCADE MVP® system reduces the time to ambulation. It was demonstrated that the median time to ambulation was 2.2 hours, which results in a 64% reduction in median time to ambulation compared to manual compression.1

Designed for EP Procedures
Ordering Information
|
AMBULATE Trial
The VASCADE MVP® system is indicated for use in catheter-based procedures6 with single or multiple access sites in one or both limbs and is an extravascular closure device to offer collagen with a dual method of action, mechanical and physiological. Using a single operator, the VASCADE MVP system is simple and easy to use and leaves no sutures or other materials behind in the vessel. The bioabsorbable thrombogenic patch expands by 13X to fill the tissue tract.7 The VASCADE MVP system allows for re-access, after 30 days, for repeat ablation procedures.
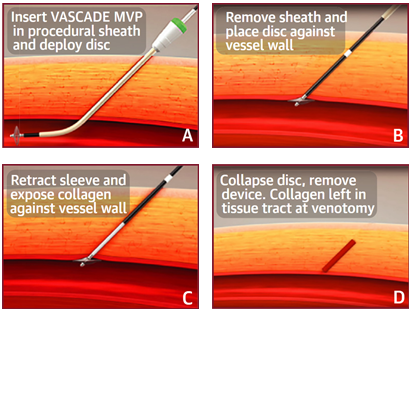
Proven by EPs
Ordering Information
|
AMBULATE Trial
|
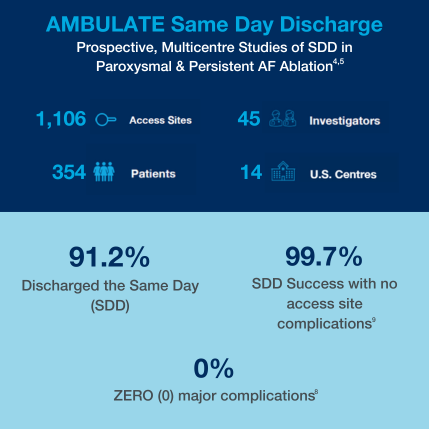
Same Day Discharge
The AMBULATE clinical studies have demonstrated 0% major complications8 in 1,223 total patients studied in 5 EP clinical trials, including 4 trials studying same day discharge paroxysmal and persistent AF ablation patients. 1-5
The AMBULATE randomised clinical trial, comparing the VASCADE MVP system vs manual compression, has demonstrated 0% major complications and significantly lower rate of minor complications. It confirmed a 63% improvement in patient satisfaction. This might be driven by the 58% reduction in opioid use and a 51% reduction in pain medication use post procedure/following ablation.1
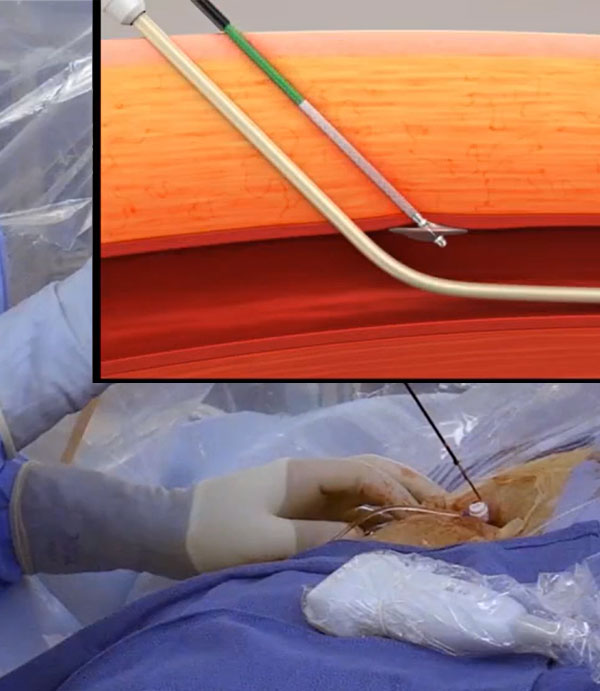
Simple and Easy to Use
✓ Single operator
✓ Indicated
• For use in 6F to 12F ID (max 15F OD) 12 cm introducer sheathsa
• For single or multiple access sites in one or both limbs
✓ 2 mechanisms of action
• Mechanical
• Physiological
✓ Extravascular design
• No permanent or intraluminal implants
• No sutures or material left in vessel
✓ Bioabsorbable and thrombogenic collagen patch
• Expands to fill tissue tract
aOverall length of the sheath (including the hub) needs to be less than 15 cm
Resource Centre
Find the information you need as you browse through helpful, on-demand information to aid in your understanding of our vascular closure solutions.
Featured Resources View all resources
Instructions for Use
The VASCADE MVP® Venous Vascular Closure System (VVCS) is intended to seal the femoral venous access site(s) by delivering a resorbable collagen patch, extra-vascularly, at the vessel puncture site to aid in achieving haemostasis.
VASCADE MVP® Brochure
The VASCADE MVP system provides rapid haemostasis for electrophysiology procedures, enabling patients to get up and moving earlier.
AMBULATE Trial
This prospective, multicentre, randomised study compared the efficacy and safety of the VASCADE MVP® system to manual compression for closing multiple access sites after catheter-based EP procedures.
AMBULATE Same Day Discharge Clinical Studies
This prospective, multicentre single arm registry evaluates procedural outcomes using the VASCADE MVP® system following atrial fibrillation interventions for patients who are discharged the same day.
Ordering Information
| Catalogue Number | Description | Order Quantity |
| 800-612C-10E | 6-12 French (Inner Diameter) | 1 Box (10 devices per box) |
| Catalogue Number | Description | Order Quantity |
| 700-500DX-05E | 5 French (Inner Diameter) | 1 Box (5 devices per box) |
| Catalogue Number | Description | Order Quantity |
| 700-580I-05E | 6/7 French (Inner Diameter) | 1 Box (5 devices per box) |
2. Al-Ahmad A, et al. Results from the prospective, multicenter AMBULATE-CAP trial: Reduced use of urinary catheters and protamine with hemostasis via the mid-bore venous vascular closure system. VASCADE MVP following multi-access cardiac ablation procedures. J Cardiovasc Electrophysiol 2021. 32(2): 191-99.
3. AMBULATE Same Day Discharge Registry Retrospective Study: NCT04538781.
4/5. Eldadah ZA, et al. Same-day discharge following catheter ablation and venous closure with VASCADE MVP: A post-market registry. J Cardiovasc Electrophysiol 2022. https://doi. org/10.1111/jce.15763. NCT04203329.
6. Catheter-based cardiac ablations requiring two or more venous access sites within the same limb. See VASCADE MVP IFU 5686 Indications for Use.
7. Data on file at Haemonetics.
8. Major venous access site closure-related complications through the follow up.
Not all products are available in all markets.
 Select Language
Select Language  English (United States)
English (United States)  English (Canada)
English (Canada)  French
French  German
German  Italian
Italian  Japanese
Japanese  Russian
Russian  Spanish
Spanish  Chinese
Chinese 

.jpg?la=en-gb&hash=C68011FE67002CA9B3D5CEF033EFE284)