Haemonetics Receives FDA Approval for Expanded Labeling of the VASCADE MVP® XL Venous Vascular Closure System
Learn more about the expanded indication here
VASCADE MVP® XL
The only extravascular venous closure system clinically proven in EP procedures using 17F OD procedural sheaths. Now with expanded indication utilizing 10-14F ID (17F max OD)
Now approved for use with:
-FARADRIVE™ Steerable Sheath
-WATCHMAN TruSteer™ Access System
-Agilis NxT Steerable Introducer, Dual-Reach™
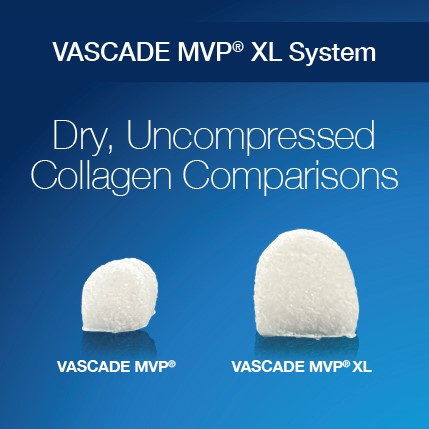
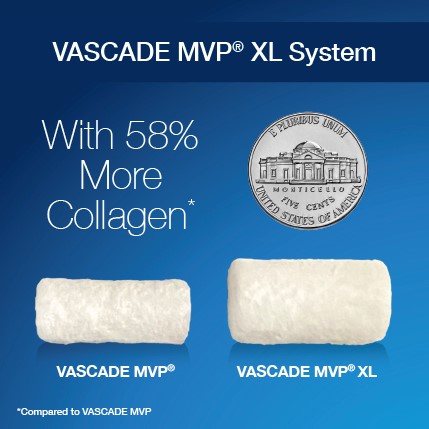
VASCADE MVP® XL venous vascular closure system is simply bigger, with 58% more collagen and a 9% larger disc compared to the VASCADE MVP system. VASCADE MVP XL is approved for the closure of femoral venous access sites during electrophysiology (EP) procedures, while reducing time to ambulation, time to hemostasis, as well as enabling same day discharge following cardiac ablation procedures, utilizing 10-14F inner diameter (17F maximum outer diameter) procedural sheaths, with single or multiple access sites in one or both limbs.
-500x574/masthead-placeholder-500-x-574/vascade-mvp-xl-hospital_masthead-images-(not-home-page)-500x574.jpg?as=0&w=641&hash=40052D23C8C9CDB8A905AEF86263122D)
VASCADE MVP® XL System
Make a selection to learn more
VASCADE MVP® XL System
Now with an expanded indication for 10-14F inner diameter (17F max outer diameter) sheaths
Ordering Information
|
AMBULATE EXPAND Trial
The AMBULATE EXPAND Trial was a prospective, single-arm, pivotal trial at 8 U.S. centers to evaluate femoral venotomy after catheter-based procedures using 10–14F ID / 17F max OD sheaths. Demonstrated safety and efficacy of the VASCADE MVP® XL venous vascular closure system in 77 patients as follows:1
- 0% major complications2
- 0% minor complications3
- Median 2.4 hours time to ambulation (TTA)4
- Median 2.3 minutes time to hemostasis (TTH)
- Median 2.7 hours time to discharge eligibility (TTDE)

Simply Bigger*
Ordering Information
|
AMBULATE EXPAND Trial
The VASCADE MVP® XL venous vascular closure system offers a 58% increase in collagen and 9% increase in disc size compared to the VASCADE MVP® system with the same simplicity and ease of use.
Discover how the VASCADE MVP® XL system can make a difference for you and your patients today.
Sharing Risks by Sharing Costs™
Ordering Information
Because Haemonetics® is so committed and confident in VASCADE MVP® XL's ability to safely and rapidly achieve hemostasis, we offer a unique value-based purchasing program called the VASCADE MVP XL Performance Guarantee. In the event of a qualified complication as the result of VASCADE MVP XL, Haemonetics will provide financial reimbursement to your hospital or practice. It’s simple, we’re dedicated to exceptional outcomes for you and your patients. We stand behind our products based on proven safety and efficacy published in peer-reviewed clinical studies.


Webinars

Resource Center
Featured Resources View all resources
Instructions for Use
The VASCADE MVP® XL Venous Vascular Closure System (VVCS) is approved for the percutaneous closure of femoral venous access sites while reducing time to ambulation, total post-procedure time, time to hemostasis, and time to discharge eligibility in patients who have undergone catheter-based procedures utilizing 10-14F inner diameter (17F outer diameter) procedural sheaths.
VASCADE MVP® XL Product Brochure
The VASCADE MVP® XL is an extravascular venous closure system designed to enable safe and efficient closure of large-bore access sites (10–14F ID/17F OD) in EP procedures such as PFA and LAAC. It is clinically proven to deliver rapid hemostasis and ambulation with 0% major and minor complications 2,3.
VASCADE MVP® XL AMBULATE EXPAND TRIAL
The AMBULATE EXPAND Trial demonstrated the safety of the VASCADE MVP® XL venous vascular closure system when compared to performance goals (PG) and clinical acceptance criteria (CAC) for closing percutaneous femoral venous access sites at the completion of catheter-based procedures utilizing 16-17F OD procedural sheaths.
Ordering Information
| Catalog Number | Description | Order Quantity |
| 700-500DX-05U | 5 French (Inner Diameter) | 1 Box (5 devices per box) |
| Catalog Number | Description | Order Quantity |
| 700-580I-05U | 6/7 French (Inner Diameter) | 1 Box (5 devices per box) |
| Catalog Number | Description | Order Quantity |
| 800-612C-10U | 6-12F Inner Diameter (15F maximum outer diameter) | 1 Box (10 devices per box) |
| Catalog Number | Description | Order Quantity |
| 800-1012XL-10U | 10-14F Inner Diameter (17F maximum outer diameter) | 1 Box (10 devices per box) |
*Compared to the VASCADE MVP system
- VASCADE MVP XL IFU 4911
- Major venous access site closure-related complications through 30-day follow up
- Minor venous access site closure-related complications through 30-day follow up
- In Per Protocol population (N=66)
 选择语言
选择语言  English (United States)
English (United States)  English (Canada)
English (Canada)  English (Other Regions)
English (Other Regions)  French
French  German
German  Italian
Italian  Japanese
Japanese  Russian
Russian  Spanish
Spanish 
